|
(Optional additional reason: fire hazard in the presence of water. Within the droplet, the hydroxide ions can move inward to react with the iron(II) ions moving from the oxidation region. Reactivity like this means the sacrificial anode would be depleted rapidly and need to be replaced frequently. This means that the lithium would react quickly with other substances, even those that would not oxidize the metal it is attempting to protect. 
While the reduction potential of lithium would make it capable of protecting the other metals, this high potential is also indicative of how reactive lithium is it would have a spontaneous reaction with most substances. In the case of iron and copper (0.34 V), iron has the smaller standard reduction potential and so corrodes (serves as the anode).Ħ. In the case of iron (−0.447 V) and zinc (−0.7618 V), zinc has a more negative standard reduction potential and so serves as the anode. The (sacrificial) anode is the metal that corrodes (oxidizes or reacts). Both examples involve cathodic protection. The cell in Figure 17.3 is galvanic, the spontaneous cell reaction involving oxidation of its copper anode and reduction of silver (I) ions at its silver cathode: cell reaction: Cu ( s) + 2 Ag + ( a q) Cu 2+ ( a q) + 2 Ag ( s) anode half-reaction: Cu ( s) Cu 2+ ( a q) + 2 e cathode half-reaction: 2 Ag + ( a q) + 2 e 2 Ag ( s. The simplest method is the use of a sacrificial anode.\begin, which appears to be able to protect all the other metals listed in the standard reduction potential table?Īnswers to Chemistry End of Chapter ExercisesĤ. The anode is the electrode at which a net oxidation reaction occurs, whereas cathodes are electrodes at which net reduction reactions occur. Earth abundant conversion cathode material iron trifluoride (FeF3) has a high theoretical capacity (712 mAh g1) and the potential to double the energy density of the current cathode material based on lithium cobalt oxide. The low DC voltage is then impressed in the area between the cathode and the anode, with the anode buried. The increasing demands from large-scale energy applications call for the development of lithium-ion battery (LIB) electrode materials with high energy density. Since electrons flow from the anode to the cathode, the structure is protected from destruction because there are no electrons lost in the process. The reaction that takes place at the cathode during the corrosion of iron is: Login. ? Direct current is impressed between the structure and the inert anode. This is a galvanic cell with zinc as the anode and iron the cathode. In the anode region, Fe is oxidized to Fe2+. Different regions on the surface of iron become anode and cathode. Zinc becomes the anode while the structure of iron becomes the cathode. The process involves electrochemical reaction. A cathode made with the best electrocatalyst from this work, tested in H2-O2, has a power density of 0.75 W cm2 at 0. The active metal becomes the sacrificial anode and the less active becomes the cathode since electrons flow from the active metal to the less active metal. ? Coupling of structures like iron with a more active metal such as magnesium or zinc. The hydrothermal reaction is carried out in a Parr reactor by mixing iron sulphate salt (FeSO4 7H2O) with phosphoric acid (H3PO4) and lithium hydroxide (LiOH) in water at about 150-200 ☌ for. There are two major methods that can be used to prevent cathodic reactions in corrosion: Neutral/basic solutions: O2 + 2H2O + 4e- = 4OH-Ĭathodic reactions can be prevented using cathodic protection methods. This is a half-cell reaction at the cathode, but the full equation contains the anodic reaction. In hydrogen revolution: 2H+ (aq) + 2e- = H2 (g) These include the reduction of the oxidation number, a condition known as reduction, and the gain of electrons at the cathode.Ĭathodic reactions can be represented using reaction equations like the following: There are several things that should be noted during the occurrence of cathodic reactions. 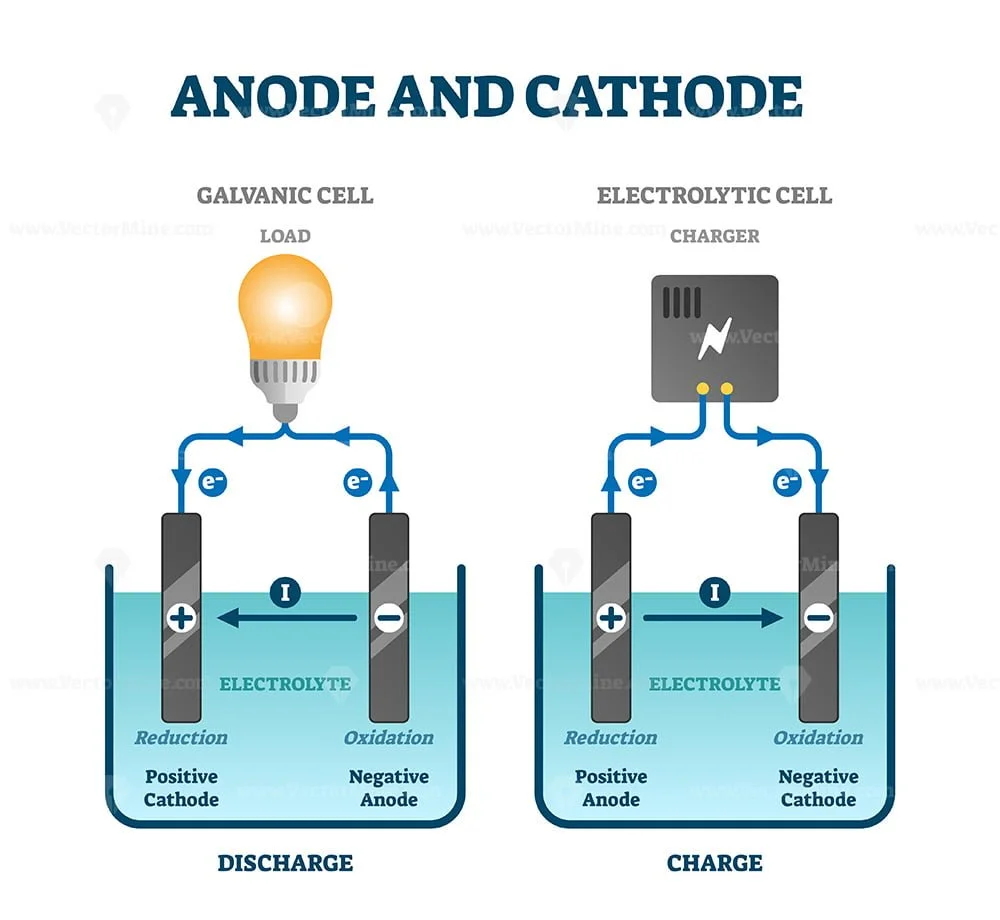
Corrosionpedia Explains Cathodic Reaction Electrolysis reactions are the basic foundations of todays modern industry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
